
The term atom originated from the Greek word atomos. His atomic model consisted of spherical shells of electrons on various states surrounding the positively charged nucleus. Bohr’s used spectral light emissions to conclude that electrons had specific energy levels, known as quantized energy levels. Niels Bohr refined the findings of Rutherford in the early 1900s. He came to his conclusion after analyzing the results of his experiment, known as the Rutherford scattering experiment, in which he found that atoms contained an extremely small, dense, and positively charged nucleus and that the area around the nucleus was mostly empty space with a few negative electrons. Thomson developed the “plum pudding model,” in which electrons were embedded in a positively charged sphere.Įrnest Rutherford developed the nuclear model of the atom in the early 1900s. He conducted further experiments to investigate the relationship between electric charge and matter. 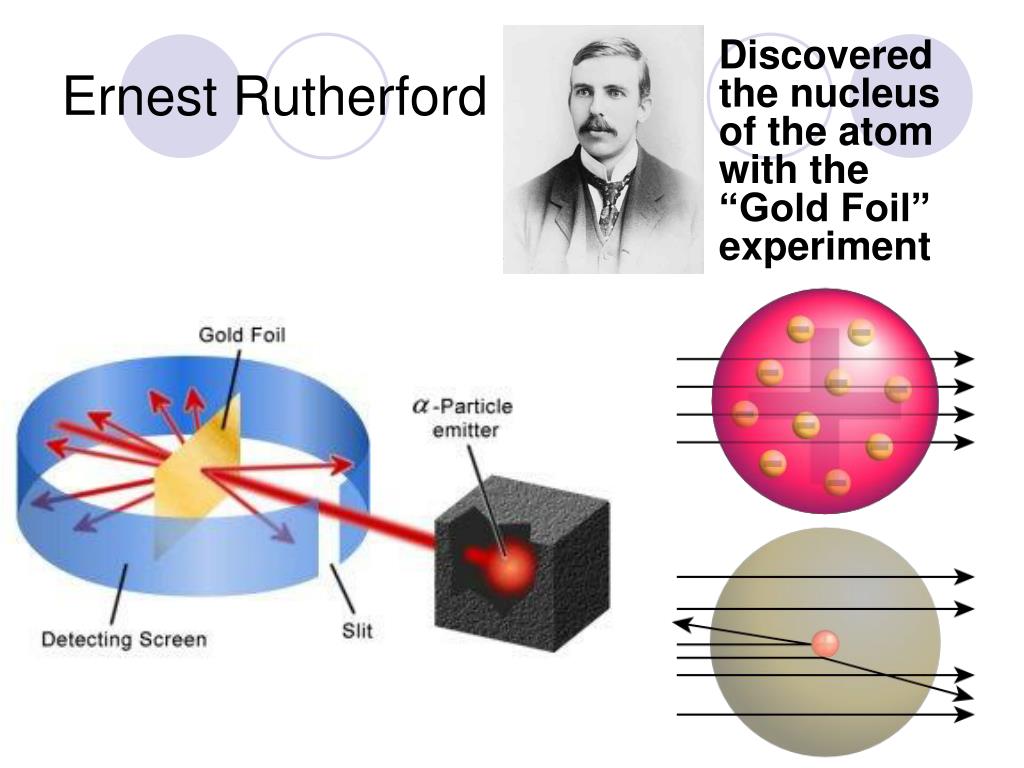
In the late 1800s he discovered that these rays were actually negatively charged particles with a charge of 1-, and that they were much smaller than atoms. Thomson experimented with particles known as cathode rays. The scientific community accepted his postulates due to his sound experimental evidence. His resulting five postulates of atomic theory helped to define the structure and nature of the atom. John Dalton extensively investigated the nature of gases in the early 1800s in order to gain a better understanding of the laws of conservation of mass and of multiple proportions. These ideas, though not scientifically tested, formed the basis for later scientists to build upon and develop modern atomic theory. A more detailed discussion of the atom will have to wait for more questions from eager Inquiring Minds readers! Be sure to send us your physics questions.The concepts of the atom and the nature of matter originated with Greek philosophers more than two thousand years ago. Since these electron clouds (10 -10 m) are 10 000 times larger than the radius of the nucleus, the atom is mostly empty space. Negatively-charged electrons orbit the nucleus in cloud-like volumes of space designated "orbitals" in quantum theory. We know today that the atom consists of a tiny (10 -14 m) nucleus containing positive charges (protons) and neutral particles (neutrons), both of which consist of more fundamental particles called quarks. Now however, he had found the nucleus of the atom, thus proving the atom had components. 
Rutherford knew from Thomson’s work that atoms contained electrons. .jpg)
An occasional alpha particle speeding directly toward such a core would be slowed, stopped, and rebound directly back due to electromagnetic forces. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you." The only way alpha particles could bounce directly back toward the "gun" that fired them was if the atom had a compact, positively charged core. In Rutherford’s words, "It was the most incredible event that has ever happened to me in my life. But to Rutherford’s amazement, Marsden found that some alpha particles came straight back after being fired at a thin gold foil. Indeed most alpha particles were scarcely deflected. Hans Geiger, Rutherford’s colleague, suggested that their student Ernest Marsden look for deflections of alpha particles through very thin foils, feeling certain that large deflections would not occur. Some radioactive substances emit alpha particles (very massive, positively charged particles) and Rutherford thought that by studying how positive alpha particles in a beam were scattered by the atoms of a material one could infer the atom’s internal structure. Ernest Rutherford (1871-1937) had been involved in studying the scattering of alpha particles by sheets of metal and mica. The major experimental indication of the atoms’ structure was Rutherford’s Scattering Experiment. In his model, the atom was a mix of equal numbers of positive and negative charges. Thomson (1856-1940), discoverer of the electron, proposed what we call the "plum pudding model" of the atom. His atom however was like a solid billiard ball. John Dalton (1766-1844), a great chemist, really started the modern atomic hypothesis. This idea of tiny, indivisible bits of matter persisted until the 1800’s. The Greek philosopher Democritus (460-370B.C.) conjectured that all things were composed of small, indivisible bits called "atoms," rendered from the Greek atomos meaning "indivisible." Democritus’ atoms came in different sizes and could combine or separate. The structure of the atom has always been one of the most intriguing areas of physics research. Who first discovered that atoms are made up of other particles and how was this found? July 2002
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
